Q: I have a validated stability-indicating assay that uses one manufacturer’s C18 column. I know of another column manufacturer where I can get an equivalent C18 column for half the price of the column stated in the method. Will changing columns invalidate the method?
A: It depends on how the method was written. If the method was written with a statement such as “use column X or equivalent,” you may be able to substitute one column for another. However, be aware that just because a column says it is C18, or because it claims to be equivalent to another, doesn’t mean that it is for all methods. You need to demonstrate column equivalency. The test is if your system suitability passes with the new column. If you get retention, resolution, and other parameters specified in your system suitability test, then you should be able to use the alternate column. I would run the test with the system suitability sample and with a few typical samples to be sure that the impurities and degradation products show up at the right retention time and concentration and the results of the analysis on the new column are equivalent to those on the old one. And be sure to document everything – as the saying goes, “if it isn’t documented, it didn’t happen.”
A good way to identify equivalent columns is to use the USP database at www.usp.org/USPNF/columns.html, which contains information to compare the selectivity of >450 reversed-phase HPLC columns. After the initial sign-in screen, you’ll see a screen with the “About the Columns Data” choices (Figure 1). Pick the last selection, “Compare Columns,” to go to the databases. There are two databases, the first called “USP Database,” and the second called “PQRI Database.” You want the second, PQRI. In the PQRI database, you’ll see a screen that should look similar to that in Figure 2. Figure 1.
Figure 1.
Then:
1. Select the column you are currently using (or that is recommended for your method) from the drop-down box in the upper left. I chose an ACE 5 C18 column as an example.
2. Make sure the text below this says “You are viewing similar columns,” and that the “View Different” button is showing. If the button says “View Similar,” click it to switch back to the equivalent column mode.
3. Just above the table, click on the appropriate check boxes for “Acids present,” “Bases present,” and indicate the pH of the separation (2.8 in this example).
4. Click the “Update” button. You will see a list of columns at the bottom of the screen with your selected column at the top. This list shows the 10 most similar columns, with the most like your selection at the top. The “F” number in the second column of data rates the similarity, and in our experience, if the value is ≤3, the columns are excellent candidates for equivalent columns. In the present example, the Develosil and Hypersil columns rate most highly, so I would choose one of these as my first choice. The column manufacturer is listed at the far right, often with a link to their website.
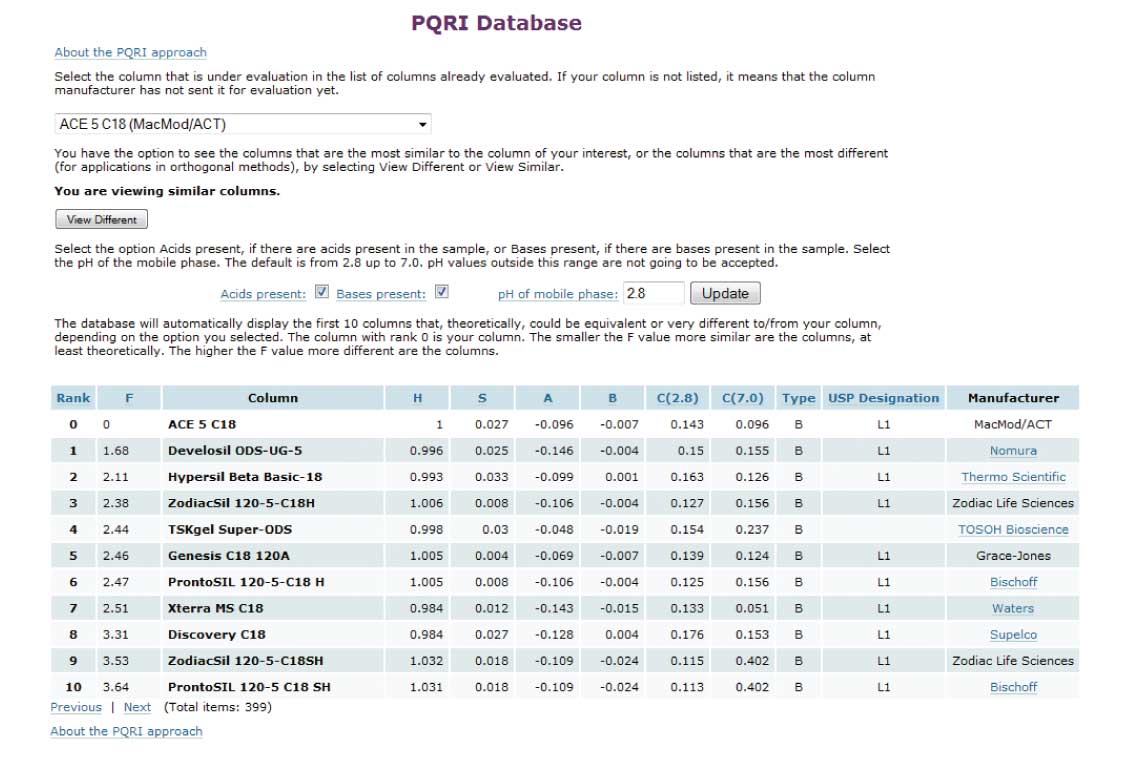 Figure 2
Figure 2
While this is not a foolproof method of identifying an equivalent column, it has been shown to be quite reliable in tests by several pharma companies [1]. Please note that this is not a guarantee of column equivalency, but greatly improves your odds of choosing an equivalent column – you still have to test the column to be sure it meets your requirements.
Reference
[1] J.W. Dolan et al., J. Chromatogr. A, 1060 (2004) 59-74.
This blog article series is produced in collaboration with John Dolan, best known as one of the world’s foremost HPLC troubleshooting authorities. He is also known for his research with Lloyd Snyder, which resulted in more than 100 technical publications and three books. If you have any questions about this article send them to TechTips@sepscience.com



