The dwell volume comprises all the HPLC system volume between the point the solvents are mixed to the head of the column. For high-pressure-mixing systems, this includes the mixer, connecting tubing, and sample loop. For low-pressure-mixing systems, we have the same components plus the volume of the pump heads and check valves. In this article let’s look at some of the problems that can be created when the dwell volume differs between two HPLC system.
A Typical Problem
Let’s consider the following scenario illustrated in Figure 1. The conditions are noted in the upper right corner: a 10-40% B gradient over 15 min at 1 mL/min on a 100 x 4.6 mm column. The method was developed and validated. Part of the system suitability requirements are that the first two peaks need to come off between 3 and 4 min and should be baseline-resolved. The method works quite well under these conditions. When the method is transferred to another HPLC system, the results shown on the bottom chromatogram are obtained. Now the first two peaks have too large of retention times and are no longer resolved to the baseline, so system suitability does not pass. What went wrong, and what can be done to correct it?
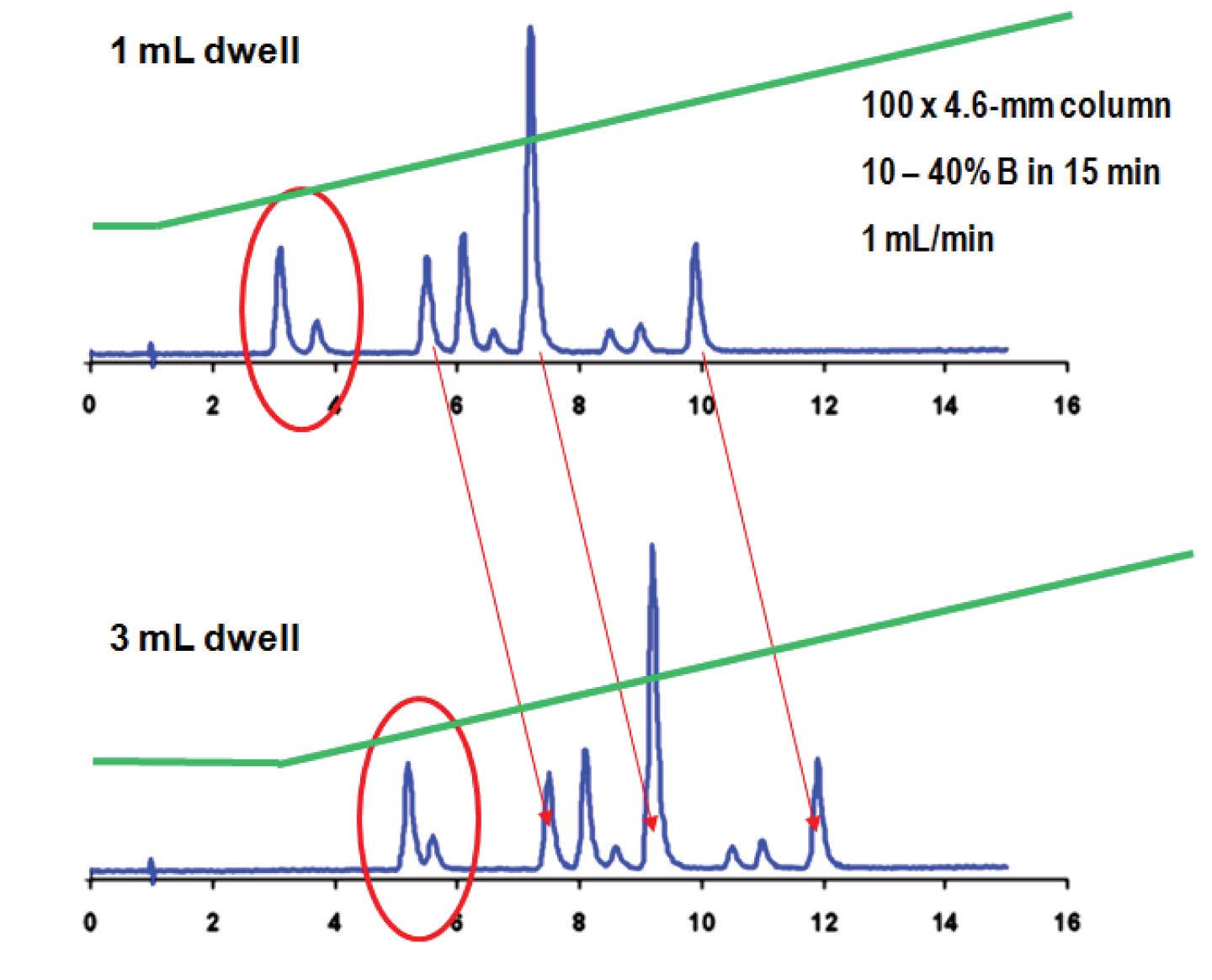
Figure 1
Different Dwell Volumes
The key difference between the two chromatograms in Figure 1 is that the HPLC systems have different dwell volumes. In the upper case, the dwell is 1 mL, whereas it is 3 mL in the lower case. Although a 10-40% B gradient was programmed, you can see in the actual gradient overlays (green) that there is a short isocratic hold at the beginning of the run. This represents the amount of time it takes to wash out the dwell volume, or the delay before the gradient reaches the column. An oversimplified description of gradient elution is that the compounds sit on the head of the column until a strong enough solvent comes along to wash them off, then they wash through the column. If this is the case, you would expect that the top system would have a delay of 1 min (1 mL / 1 mL/min), whereas the bottom one would have a delay of 3 min (3 mL / 1 mL/min), corresponding to the isocratic hold in the green gradient overlays. Thus, all the peaks would be expected to come off the column (3 min – 1 min) = 2 min later in the lower case. This is true for the peaks at the end of the chromatogram, but not for those at the beginning. The reason is that early-eluted peaks don’t really sit still until the gradient hits the column, but migrate a bit under the initial isocratic conditions. Therefore the first two peaks migrate for 1 min isocratically plus ≈ 2 min under gradient conditions in the upper case, whereas there are 3 min of isocratic migration and ≈ 2 min gradient migration in the lower case. This is enough difference that the peaks are not only offset, but also spaced a bit differently between the two systems.
Differences in dwell volume are the most common reason that gradient methods are hard to transfer from one system to another and why it is difficult to reproduce published methods. Because the dwell volume is so important, it is wise to include the dwell volume as part of the system description when documenting gradient methods. In the next few installments of HPLC Solutions we’ll look at how to measure dwell volume, how to compensate for dwell volume differences, and additional problems that may be encountered with gradients.
This blog article series is produced in collaboration with John Dolan, best known as one of the world’s foremost HPLC troubleshooting authorities. He is also known for his research with Lloyd Snyder, which resulted in more than 100 technical publications and three books. If you have any questions about this article send them to TechTips@sepscience.com




