In this article we discuss issues related to inlet and liner activity.
Many of the weaknesses inherent in gas chromatography are related to problems with the transport of sample into and through the inlet. Of these problems, sample loss (the inverse of recovery) and chemical changes (e.g., decomposition) are related to inlet activity, and much of that is dominated by the inlet liner.
The cool on-column inlet is considered the gold standard for representative sampling because of its low (or absence of) discrimination and minimal interaction with samples. However it is probably the trickiest injection technique to use on a routine basis and exposes the column directly to sample, large volumes of condensed solvent and sample matrix, lowering column lifetime. It also is less rugged, requires more operator expertise and more sample cleanup than do hot or programmable split/splitless inlets. The following discussion is relevant, then, to split/splitless style inlets (hot and programmable).
Inlet activity can manifest in several ways:
- Irreversible adsorption (partial or total losses)
- Chemical changes (decomposition, polymerization, rearrangement)
- Peak tailing
My experience has taught me that there are too many variables involved with inlet activity for all to be controlled. In fact, what reduces activity toward some compounds can increase activity toward others. In addition, the nature and extent of activity changes with every injection of sample or even clean standards.
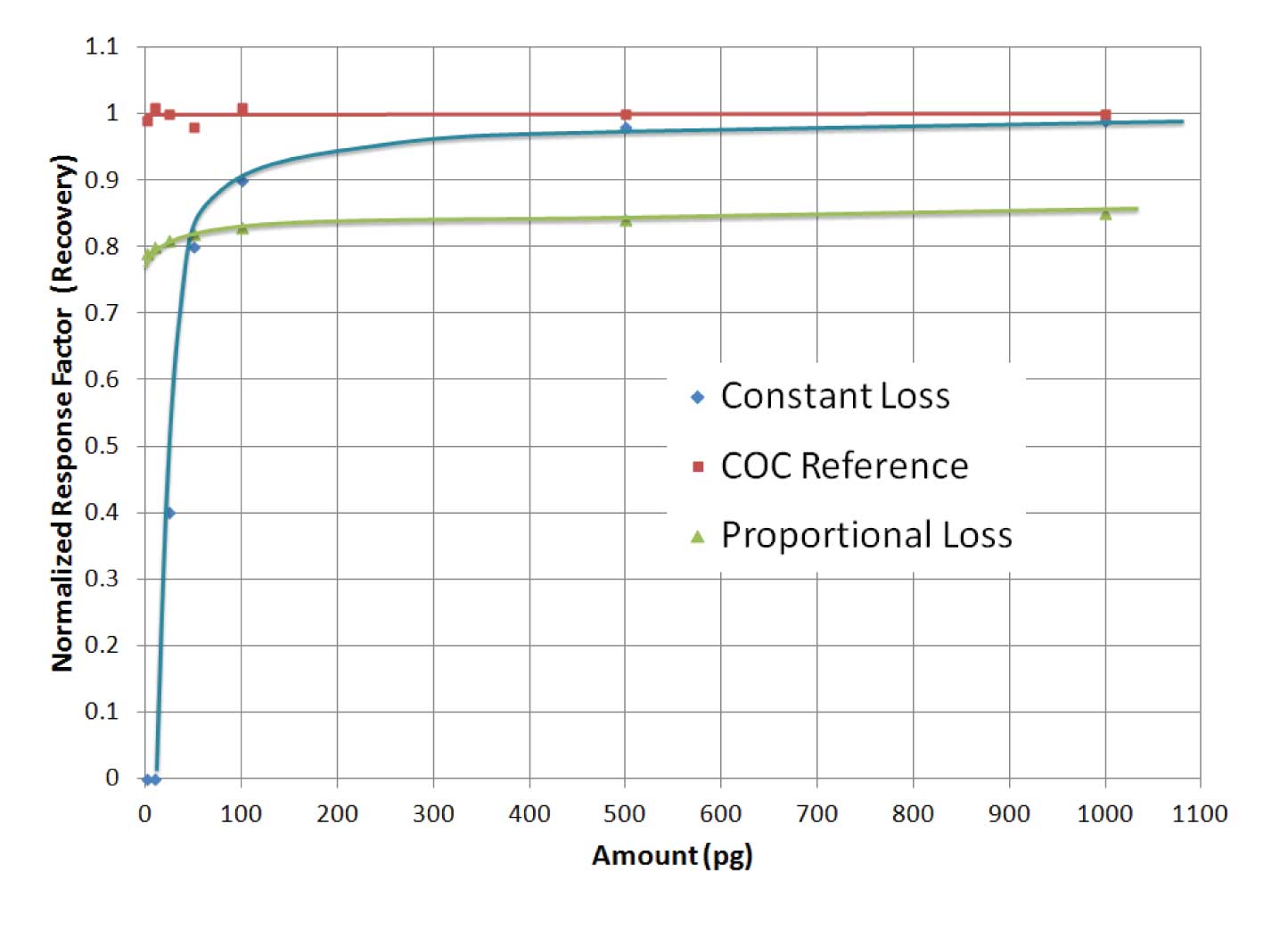
Figure 1: Response factors (normalized to cool on-column injection) as a function of injected amount. Blue normalized response factor curve A shows a dramatic loss of analyte below 100 pg, indicating a constant loss of material. Green normalized response factor curve indicates that there is a proportional loss.
The stronger the polarity of the analyte (acid/base > hydrogen bond acceptor/donor > permanent dipole > aromatic > unsaturate), the more dramatically it will be influenced by inlet activity. The more unstable its chemical bonds, the more likely it is to decompose.
The higher the inlet temperature, the more pronounced might be the decomposition but the less pronounced might be the adsorption (i.e., bad for some things, good for others). This is one reason it is impossible to have a general set of conditions that is best for everything. Optimal GC inlet conditions are very specific to the sample, solvent, inlet design, choice of column and flow rates, etc. Inlet variables should be investigated and optimized after other method variables have been optimized and then held constant.
Inlet weldment (the permanent part of the inlet, the heated metal block) activity can come from many sources. These include remnants of the manufacturing process, unclean parts (machining lubricants, debris such as metal filings, “hot spots” such as nicks or scratches in the metal, rough metal surfaces with high surface areas, and low grade steel iron or even rust at the surface, carbon).
Proper manufacturing processes can reduce these sources of activity. Inlet weldment deactivation through coating processes such as those employed by Silco Tek (Bellefonte, Pennsylvania, USA) can further reduce activity; however activity can never completely be removed, which is why all heated inlets use liners.
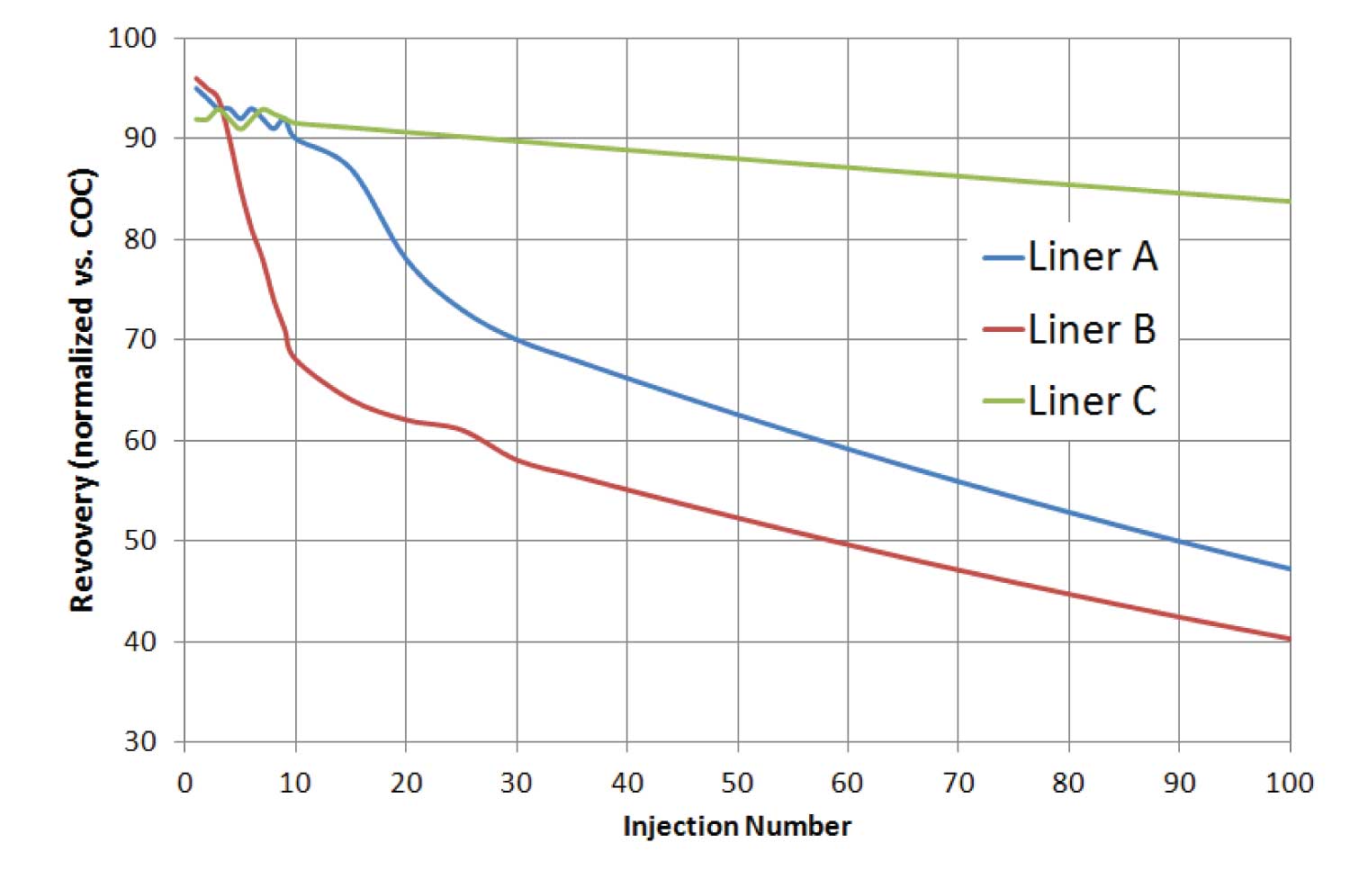
Figure 2: Recovery curves as a function of injection number. Liner C shows much more stability of its inertness than the other two. Liner B, an undeactivated glass liner, showed good initial inertness but quickly degraded after only a few injections.
A common perception is that sample will never be exposed to the inlet weldment because sample vapors will be fully contained within glass or quartz inlet liners. But unless the column is intimately mated to a taper at the bottom of the liner some sample will blow around the column inlet, diffuse around the outside of the liner and thereby be exposed to the weldment. The wider the taper at the bottom of the inlet and the looser the liner in the inlet weldment, the more sample will be exposed to the liner and the longer the time of contact.
Sample outside the liner can also be exposed to activity from the bottom plate (usually replaceable), debris at on the bottom plate, and ferrule material (especially graphite) that is exposed at (or extruded into) the inlet base.
A common way to examine total inlet activity (liner + any exposure to weldment or bottom seal) is to compare recoveries (responses) of activity probes injected into a given inlet to those using a cool on-column inlet, with all other GC system components in the sample path the same. Alternatively, one can normalize responses of the active analytes to analytes that are known to have no activity issues, such as saturated hydrocarbons of similar volatilities as the active analytes.
Figure 1 shows the normalized responses of a couple active analytes in an active system relative to cool on-column injection. Two general types of activity are evident; constant loss and proportional loss. Irreversible adsorption (or very slow desorption from active sites), for example, is often observed as a constant loss of analyte. Response factors “fall off the table” at low loadings. One might think of this as a fixed number of hot spots (active sites) that can be masked (titrated) by a given amount of active analyte. It could also happen if a fixed amount of analyte decomposes. Analyte recovery is almost quantitative above the fixed amount. At higher amounts of analyte, the constant loss becomes insignificant and can go unnoticed, which is not a problem if your analytical range of interest spans only high concentrations. The analytical range of an analysis is limited on the low end by this constant loss.
Proportional losses usually emanate from a catalytic or thermal mechanism whereby a constant proportion of sample is lost. In this case, response factors are more-or-less constant across the analytical range, although sensitivity is reduced.
Many people incorrectly measure inlet activity using only a few injections on a new liner. However, that can be misleading. As illustrated in Figure 2, activity changes with time. This point is extremely relevant when testing and evaluating efficacy of a given liner deactivation as well as when trying to choose the best liner for large sample sequences. I have seen bare glass liners that are more inert than deactivated liners – for the first few injections. I have evaluated deactivation processes that yielded liners with excellent inertness for 10-15 injections, after which their activity geometrically increases. With most laboratories doing routine analyses, it is reasonable to expect that a given liner’s inertness should last for at least one full tray of samples (50, 100, 150 injections). It would be nice if liner manufacturers presented performance data for more than a few injections, but few do so.
Based on the above, it is reasonable to keep the following questions in mind when interpreting recovery data as a measure of liner inertness:
- Is the test probe’s chemical structure and lability (opposite of stability) similar to that of active components in your samples?
- How close to the low end of your method’s analytical range was the injected concentration of the test probe(s)?
- What is the ruggedness of the deactivation? How many injections were made and at what temperature during the activity test?
It is best to measure inlet activity with the same compounds you wish to analyse and under the same conditions as your analysis. However, when trying to choose between commercial liners based on manufacturer provided data, this is rarely practical, so one must rely on results based on manufacturer selected probes. Focus on the probes that most closely resemble your sample components of interest.
It is also important to be cognizant of the amount of analytes (probes) that were injected. It can be deceiving to state “amount on-column” because injecting a higher concentration of probe into an active inlet with a constant loss, and then splitting in order to get lower amounts “on column” masks the constant loss. This is only a concern if the split injection technique is used for the activity tests.
This blog article series is produced in collaboration with Dr Matthew S. Klee, internationally recognized for contributions to the theory and practice of gas chromatography. His experience in chemical, pharmaceutical and instrument companies spans over 30 years. During this time, Dr Klee’s work has focused on elucidation and practical demonstration of the many processes involved with GC analysis, with the ultimate goal of improving the ease of use of GC systems, ruggedness of methods and overall quality of results. If you have any questions about this article send them to techtips@sepscience.com



