Having trouble separating compounds, and wondering what is the single most important parameter to change? Questions over characterizing high molecular weight compounds such as oligonucleotides? Tackle column selection like an expert by registering for the LC Learning Series, a free resource provided by Separation Science and PerkinElmer. Includes a high-resolution version of the HPLC column selection poster pictured below!
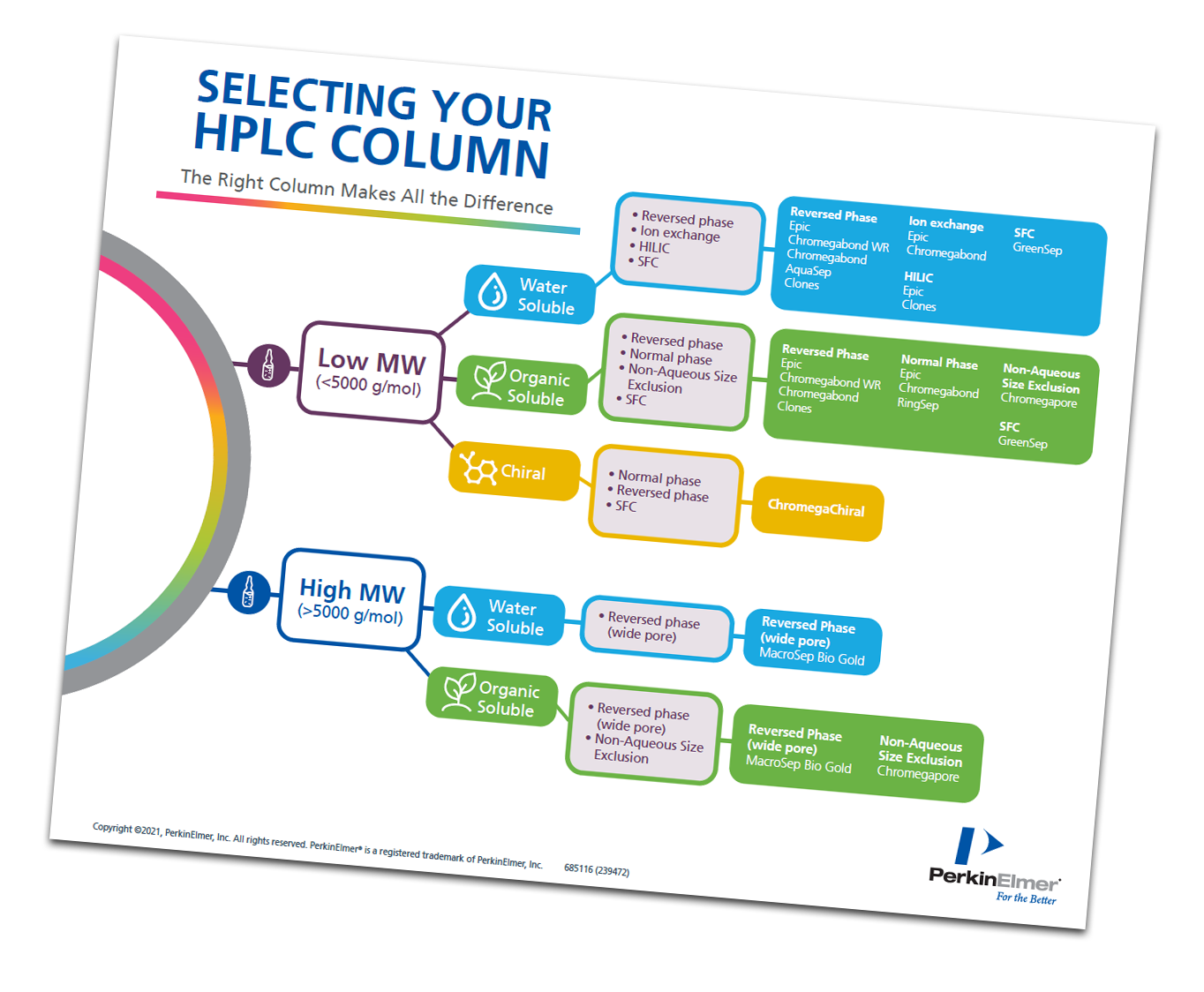
The LC Learning Series aims to help researchers simplify HPLC and UHPLC analysis through self-paced video tutorials. In tutorial 2, "LC Column Selection" Matthew Przybyciel, a Product Development Leader at PerkinElmer, takes viewers through both the fundamental elements and cutting-edge technology in HPLC column choice, including:
1. Stationary Phase Selection
- Complex analytes: Analytes are becoming more polar. While C18 reverse phase remains popular, high aqueous and HILIC phases offer alternatives for polar compounds.
- Faster Analysis: Speed matters. Researchers seek ways to reduce runtime while maintaining column longevity.
- Alternative phases beyond C18: Fluorinated phases such as pentafluoro phenyl are increasingly tackling complex natural mixtures.
- Molecular weight matters:
< 5000 Molecular Weight: Consider solubility. C18 suits water-soluble compounds; other options exist for organic-soluble or chiral molecules.
> 5000 Molecular Weight: Choose based on solubility; reverse phase chromatography remains a top choice. - Versatility matters: Ether-linked C18 handles polar and organic compounds alike, making it a smart choice for applications such as caffeine separation in beverages.
2. Particle Size
- Preparative chromatography: Use larger particles (5 to 20 Micron).
- Standard HPLC: Opt for particle sizes from three Micron to 10 Micron.
- Ultra High-Performance LC: Achieve maximum efficiency with 1.8 Micron particles.
- Matching the particle size to your HPLC system's capabilities is vital.
- Smaller particles offer advantages like enhanced efficiency, higher sensitivity, faster analysis, increased peak capacity, and reduced flow rate influence. However, they come with higher back pressure and may require system adjustments.
3. Pore Size
- Balancing act: Surface area and pore size have an inverse relationship, and most interactions between analytes and the stationary phase occur within these internal pores.
- Pore pitfalls: Too small or too large?
Small Pore Particles (60-164 Angstrom): Typically not a consideration, but they can offer unique separation characteristics in specific cases.
Large Molecules (Over 5000 Molecular Weight): For proteins, large polypeptides, or oligonucleotides, matching pore size to molecule size is crucial. Larger molecules can't enter small pores but thrive in larger ones.
4. Column Dimensions
- Internal diameter (ID): Larger diameter columns (≥4 mm) excel in sample loading capacity and standard HPLC compatibility but consume more solvent, offer lower sensitivity, and lack LC-MS compatibility. Narrower bore columns (2-3 mm ID) reduce solvent use, boost sensitivity, and align with LC-MS but hold less sample and require instrument optimization.
- Sensitivity and solvent economy: Narrow bore columns shine in sample sensitivity, with a 2.1 mm ID column offering over five times the sensitivity of a 4.6 mm ID one. They also save significantly on solvents – switching to a 2.1 mm ID column can slash solvent consumption by 80%.
The Presenter Matthew Przybyciel, Ph.D., is a Product Development Leader at PerkinElmer
Matthew Przybyciel, Ph.D., is a Product Development Leader at PerkinElmer
Matthew has a Ph.D. in Analytical Chemistry from the University of South Carolina. In the late 90s he worked in ES Industries where he has successfully developed and commercialized several chromatographic stationary phases and columns for HPLC and SFC. In 2021, ES Industries became part of PerkinElmer, Inc.

Now that you’ve learned about how to choose the perfect LC column, be sure to check out the other tutorials:
Tutorial 1: Introduction to HPLC
by Pamela A. Perrone, Ph.D., Global Support and Trainer, PerkinElmer
This presentation is the first in a series that will introduce you to the most widely used analytical technique in the world – high performance liquid chromatography (HPLC). HPLC is a separation technique that allows you to separate, identify, and quantitate the components in a sample. The objective of this session is to provide you with an entry level understanding of this technique. Topics include chromatographic theory, the different modes of HPLC, HPLC instrumentation, and where HPLC is used.
Tutorial 3: Choosing the Right HPLC detector
by Adrian Garcia Sega, Application Scientist, PerkinElmer
This session explores the most common detectors for high performance liquid chromatography (HPLC). The presentation will focus primarily on optical detection, such as UV/Vis, photodiode array, fluorescence, and refractive index detectors, however other HPLC detectors will also be considered. The objective of this presentation is to provide a basic understanding of the theory of operation, advantages and disadvantages of these detectors, and what to consider when choosing a detector for a particular analysis. Common application examples are given.
Tutorial 4: Basics of HPLC Troubleshooting
by Jamie Foss, Product Manager - LC, PerkinElmer
Presented in a Q&A format, this session of our 'LC Learning Series' explores some of the most common issues/problems encountered in the HPLC lab. As the most important diagnostic tool for HPLC is monitoring the pump pressure, several examples of pressure issues will be discussed. Other problems, such as retention time and peak area irreproducibility and detector baseline noise or drift, are presented. The aim of this session is to provide some basic troubleshooting fundamentals to build confidence in recognizing when issues are occurring and how to resolve them to improve instrument uptime and laboratory productivity.




